


Solution to Damashek's Riddle

A ROS rich environment is not good for ribosomes


Solution to Damashek's Riddle
WELCOME TO THE DINMAN LAB
My laboratory studies three distinct yet overlapping fields of study: virology, ribosome structure/function relationships, and regulation of gene expression.
Regulation of Gene Expression
Since "biological systems tend to use whatever works", there is no reason to believe that programmed ribosomal frameshifting is exclusively utilized by viruses. Based on this philosophy, we are pursuing a bioinformatic program designed to identify functional programmed -1 ribosomal frameshift signals in the genomic databases. This effort employs a combination of computational, DNA microarray, and traditional "wet lab" approaches. We have found that programmed ribosomal frameshift signals can act as mRNA suicide elements, suggesting that PRF is used to post-transcriptionally regulate the abundance of specific mRNAs and their encoded protein products. The reverse side of this coin is the question of how viruses have evolved to circumvent this regulatory mechanism, allowing them to utilize programmed ribosomal frameshifting without having their mRNAs degraded.


Virology
The maintenance of correct translational reading frame is fundamental to the integrity of the protein synthetic process, and ultimately to cell growth and viability. Despite this, it has been demonstrated that certain viruses utilize specific signals on their mRNAs that induce elongating ribosomes to shift reading frame. The highly controlled efficiencies of PRF events ensure that the proper stoichiometric ratio of viral structural to enzymatic proteins are available for viral particle assembly. Changing frameshifting efficiencies alters this ratio, preventing proper viral particle assembly and interfering with virus propagation. Thus, programmed ribosomal frameshifting presents a promising new target for anti-viral pharmacological intervention. We are characterizing a series of yeast mutants and drugs in order to identify new targets for antiviral therapies. We are also working to create a reverse genetic system for a dsRNA virus of yeast.
Ribosome Structure & Function
One important function of the ribosome is to faithfully maintain translational reading frame. Viral mRNA signals that abrogate this function by programming ribosomes to shift frame have proved to be of tremendous utility in elucidating the molecular mechanisms underlying this essential task. The newly available atomic resolution structures of ribosomes mark a critical milestone in the quest to link ribosome structure with function, and our studies on PRF have begun to link ribosome structure with translational frame maintenance. We have shown that both the biophysical interactions between ribosomal proteins rRNAs and tRNAs, and the biochemical properties of ribosome-associated enzymatic activities are both important for proper reading frame maintenance. On a broader scale, our work also is consistent with the hypothesis that communication between the different functional centers of the ribosome is critical for coordinating ribosome structure with its various functions. Of particular interest, recent structural analyses of mutants that we had previously identified as affecting frameshifting reveals that they correspond to critical points of contact between specific ribosomal components. This positions us for to conduct reverse genetic studies linking ribosome structure with function.
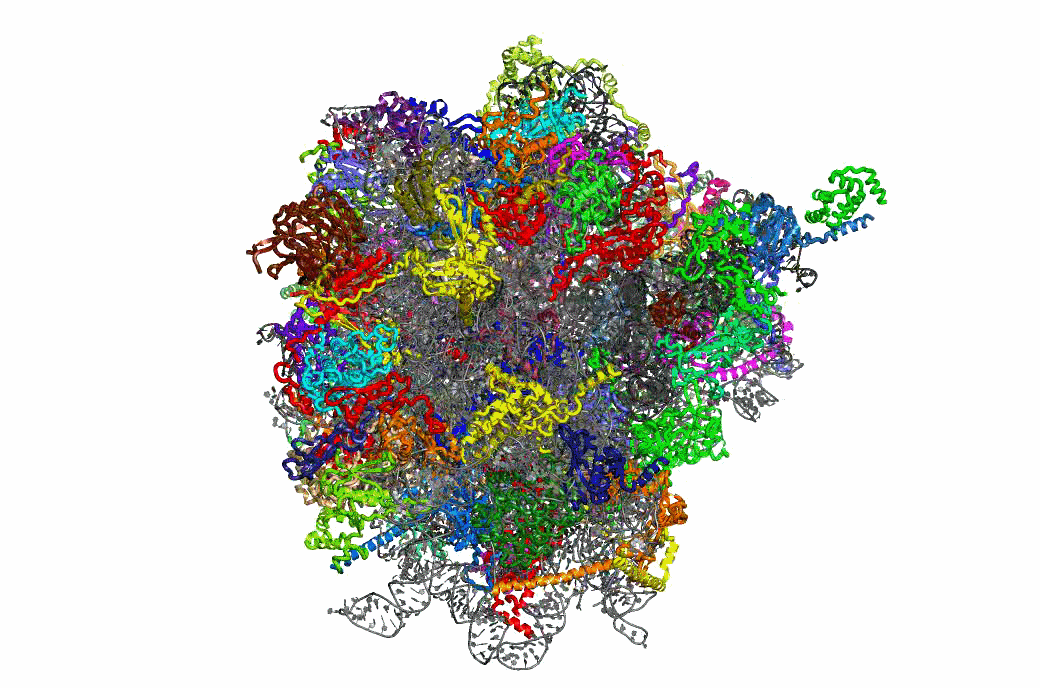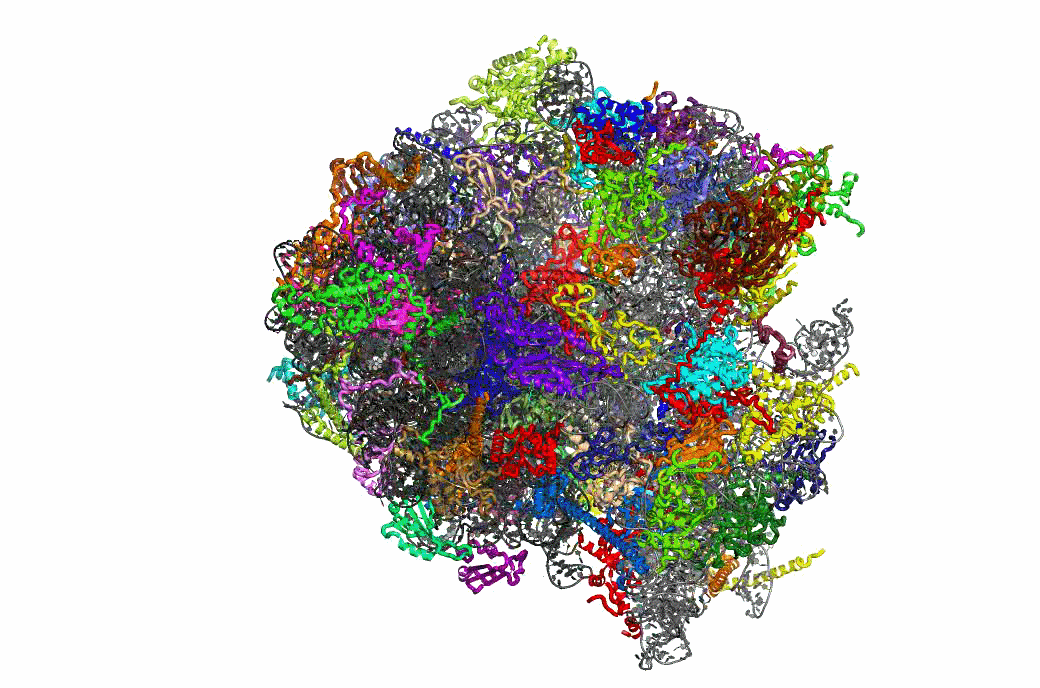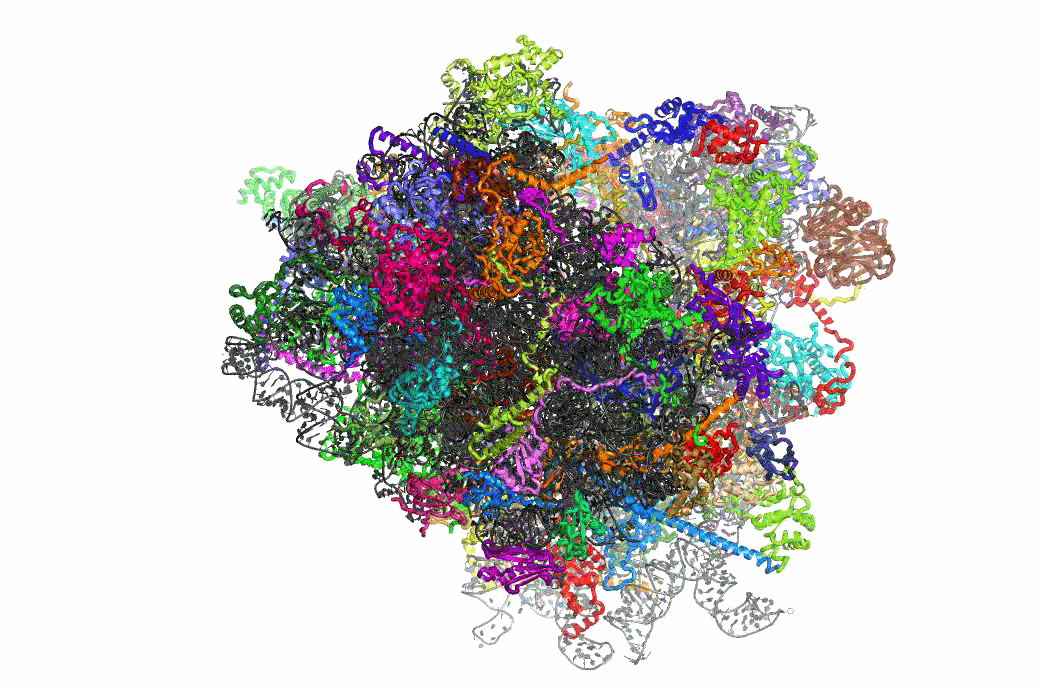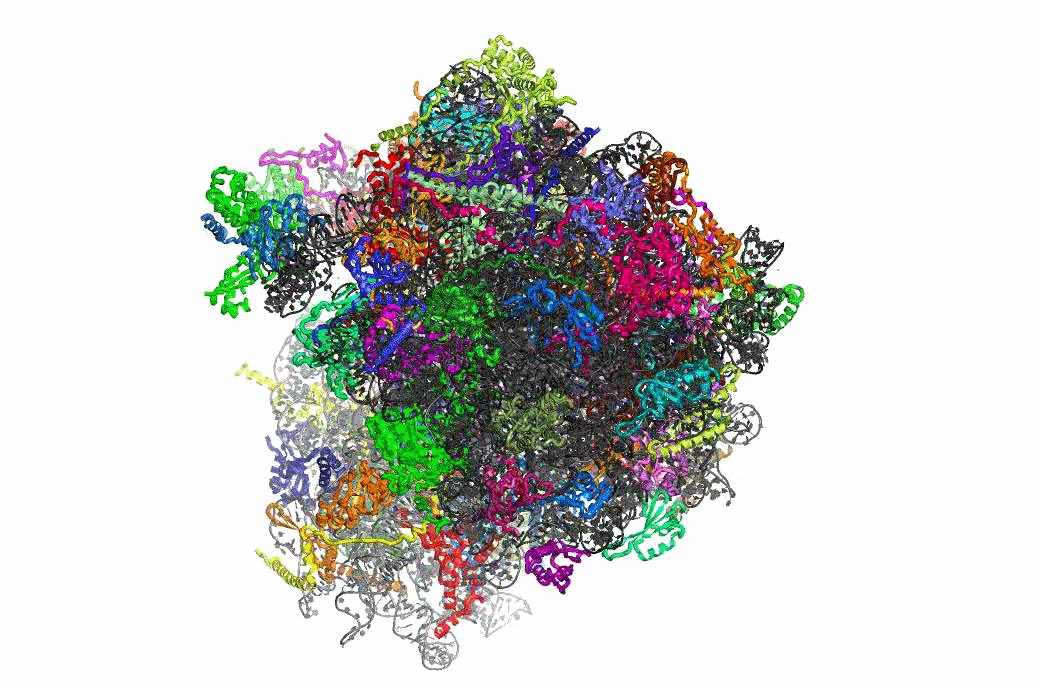
The PRFdb
The Programmed Ribosomal Frameshifting Database is now publicly available.
http://dinmanlab.umd.edu/prfdb/ or http://prfdb.umd.edu
CONTACT US
4062 Campus Drive
College Park MD 20742
Office: 301 405 0918
Lab: 301 405 1758
FAX: 301 314 9489
email: dinman@umd.edu
Useful Links:
| UMD Home | Directories | Search | Admissions | UMD Calendar |
| College of Computer Mathematical and Natural Sciences | CBMG
